A new study reveals that the assembly of amyloid proteins—central to Alzheimer’s disease—can be strongly influenced by the spin orientation of electrons on magnetised surfaces. Researchers found that the direction of magnetisation, together with the protein’s molecular “handedness” (chirality), led to dramatic differences in the number, length, and structure of amyloid fibrils formed. The phenomenon points to a direct role for electron spin, under the mechanism of Chiral-Induced Spin Selectivity (CISS), in dictating how these disease-related proteins self-assemble—a physical factor that could open new routes to controlling or interrupting amyloid build-up in the brain.
How Spin Shapes Fibril Growth
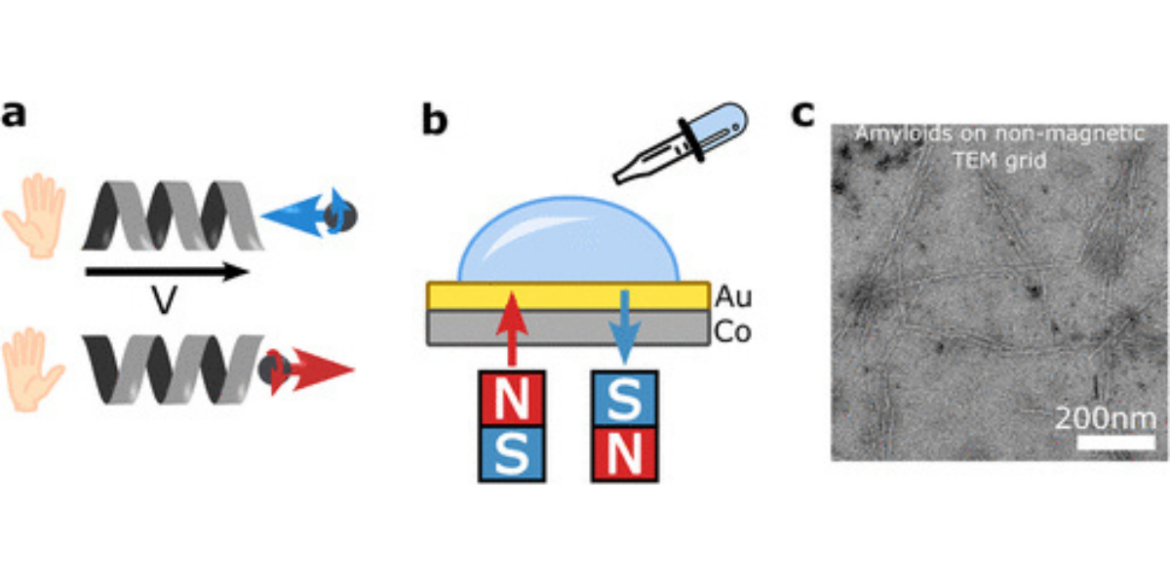
At the centre of this research is amyloid-beta (Aβ₁–₄₂), a protein fragment that forms sticky plaques in Alzheimer’s patients. By studying these peptides on magnetised surfaces, the team discovered that aligning the surface’s magnetic field in one direction produced nearly twice as many amyloid fibrils—some as much as 20 times longer—compared with the opposite direction. When using a peptide with reversed chirality, the patterns also reversed, underscoring a clear spin-dependent effect. These behaviours align with the CISS phenomenon, where chiral molecules interact differently with electrons according to their spin direction, a process now emerging as significant in biology as well as physics.
Implications for Neurodegeneration
The findings suggest that electron spin orientation can do more than subtly tweak molecular processes—it can actually steer the assembly of disease-related proteins. Using advanced imaging and spectroscopy, the researchers found not only quantitative but also structural differences in amyloid fibrils, depending on the surface’s spin alignment. These physical properties offer new ways of thinking about—and possibly intervening in—the aggregation underlying Alzheimer’s and related diseases.
Toward Future Applications
Although still basic research, the insights offer promise for future therapies. The team proposes that spin-polarised materials, such as engineered nanoparticles or bespoke membranes, might one day selectively disrupt the formation of amyloid clumps linked to neurodegenerative disease or even conditions like dialysis-related amyloidosis.
“Our work adds a new tool to understand—and perhaps eventually control—protein aggregation in diseases like Alzheimer’s,” said study co-author Prof. Yossi Paltiel.
The study, “Controlling Amyloid Assembly Dynamics Using Spin Interfaces”, was published in ACS Nano. The research was led by Yael Kapon of the Hebrew University of Jerusalem’s Institute of Applied Physics, under the supervision of Prof. Yossi Paltiel, and in collaboration with Prof. Ehud Gazit (Tel Aviv University), along with researchers from institutions in Israel and Poland.
You can read the full article HERE
Researchers:
Yael Kapon, Dror Merhav, Gal Finkelstein-Zuta, Omer Blumen, Naomi Melamed-Book, Yael Levi-Kalisman, Ilya Torchinsky5, Shira Yochelis, Daniel Sharon, Lech Tomasz Baczewski, Ehud Gazit2, Yossi Paltiel
Institutions:
- Institute of Applied Physics, The Hebrew University of Jerusalem
- The Shmunis School of Biomedicine and Cancer Research, George S. Wise Faculty of Life Sciences, Tel Aviv University
- Institute of Chemistry, Hebrew University of Jerusalem
- Bio-Imaging Unit, The Alexander Silberman Institute of Life Science, The Hebrew University of Jerusalem
- The Harvey M. Krueger Family Center for Nanoscience and Nanotechnology, The Hebrew University
- Institute of Physics, Polish Academy of Sciences, Warsaw
