A new research suggests that turning down an overactive immune sensor may help restore tissue health in severe genetic disorders, offering a fresh perspective on ageing and DNA damage.
Title Image: Dr. Marva Bergman (left), and Prof. Itamar Harel. Credit: Alon Harel-Katz
The human immune system is designed to detect and destroy viral threats. But sometimes this defence mechanism can misfire. When fragments of the body’s own damaged DNA are mistaken for invading viruses, the result is a powerful inflammatory response that can harm the very tissues it is meant to protect.
Now, an international team led by Dr. Marva Bergman and Prof. Itamar Harel at the Hebrew University, in collaboration with Prof. Yehuda Tzfati, Prof. Ido Ben-Ami of the Hebrew University and Shaare Zedek Medical Centre, and Prof. Bérénice Benayoun of the University of Southern California, has identified this misdirected immune response as a key driver of tissue degeneration in severe, rapid-ageing disorders. By reducing this false alarm, the researchers were able to improve function across multiple biological systems.
The findings focus on rare DNA damage-repair (DDR) syndromes such as Ataxia-Telangiectasia (A-T) and Bloom syndrome. In these conditions, the machinery that normally repairs everyday DNA damage is impaired, leading to genomic instability, neurodegeneration, cancer susceptibility and premature ageing.
For decades, scientists believed that the accumulation of unrepaired DNA was the main cause of cellular decline. This study challenges that idea.
“Our results show that the damage is not acting alone,” said Prof. Harel. “It is the body’s response to that damage — an exaggerated, chronic inflammatory reaction — that drives much of the degeneration.”
When DNA repair fails, fragments of DNA can leak into the cell’s cytosol, where they activate a molecular sensor called cGAS. This pathway usually detects viral DNA, but it cannot reliably distinguish between foreign and self-derived fragments. The result is a sustained, sterile inflammatory response that damages tissues.
The researchers also found a second, unexpected role for cGAS. In addition to triggering inflammation, it can enter the cell nucleus and directly interfere with DNA repair. This dual function makes it both a defender under normal conditions and a potent driver of damage when the system is overwhelmed.
To test whether moderating this response could alter disease progression, the team used a fast-ageing vertebrate model that allows rapid assessment of ageing-related processes. When cGAS activity was reduced, key disease features — including neuroinflammation, tissue degeneration and loss of reproductive capacity — were significantly improved.
“We were not just slowing decline,” said Dr. Bergman. “We saw broad restoration of tissue function. It suggests that the body can cope with more DNA damage than we assumed, if the inflammatory response is kept in check.”
The implications for treatment are important. Rather than trying to repair every DNA lesion, future therapies could focus on how the body responds to damage. However, the researchers caution that cGAS also plays a critical role in antiviral defence, so any treatment will need to selectively reduce harmful activity without weakening immunity.
Beyond rare genetic disorders, the findings may also be relevant to age-related diseases, where chronic inflammation and genomic instability often occur together.
Complementary studies from the group also point to the way fundamental biological programmes, such as reproduction and developmental timing, intersect with ageing and lifespan. Together, this work suggests a broader principle: the same systems that support early-life fitness may also help define the limits of long-term health.
The researchers note that reversing severe disease processes is not the same as slowing the intrinsic pace of ageing. Even so, by showing how the body’s own alarm system can contribute to decline, the study opens a promising new path for treating some of the most challenging degenerative conditions.
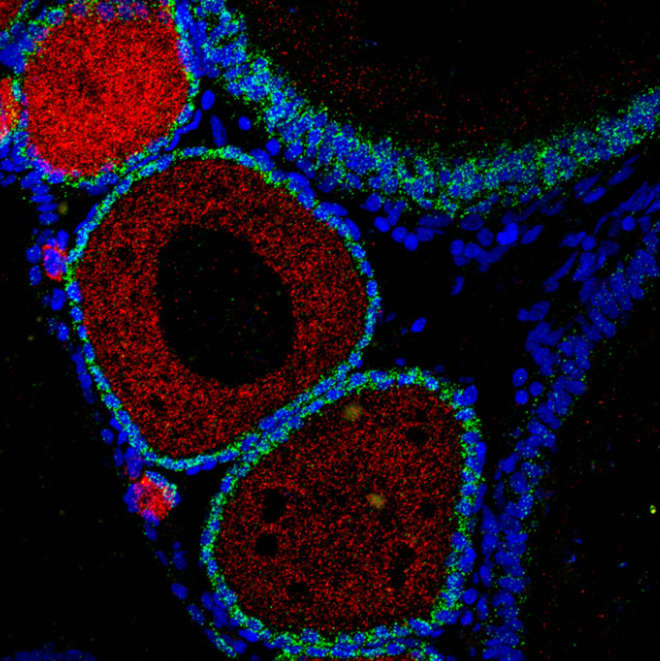
Reversing hallmarks of rapid aging by silencing a cellular alarm system – single-molecule fluorescent in-situ hybridization (smFISH) in killifish female ovaries, visualizing immature oocytes (ddx4) in red, and for granulosa cells (lhr) in green. Nuclei are visible in blue. Credit: Eitan Moses
The research paper titled “A dual role for cGAS in shaping cellular and organismal responses to genomic instability” is now available in Genes & Development and can be accessed at https://genesdev.cshlp.org/content/early/2026/04/13/gad.352760.125
Researchers:
Marva Bergman, Uri Goshtchevsky, Tehila Atlan, Gwendoline Astre, Ryan Halabi, Hosniyah El Ayoubi, Eitan Moses, Aaron J.J. Lemus, Bérénice A. Benayoun, Yehuda Tzfati, Ido Ben-Ami, Itamar Harel
Institutions:
- Department of Genetics, the Silberman Institute, the Hebrew University of Jerusalem
- Department of Obstetrics & Gynecology, the Eisenberg R&D Authority, Shaare Zedek Medical Center and Faculty of Medicine, The Hebrew University of Jerusalem
- Leonard Davis School of Gerontology, University of Southern California
- Molecular and Computational Biology Department, USC Dornsife College of Letters, Arts, and Sciences, Los Angeles